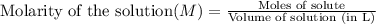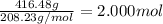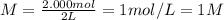Chemistry, 30.06.2019 19:00 mydoggy152
What is the molarity of 416.48 g bacl2 dissolved in 2 l of water? ! a) 1 m b) 104.12 m c) 2 m d) 208.24 m

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Select the correct answer from each drop-down menu. daniel and sanya are scientists. daniel is studying whether the increasing frequency of tropical storms is affecting coastal erosion. sanya is investigating whether the discharge from industrial plants has any impact on the ph concentration of freshwater swamps in the surrounding area. which fields of science are daniel’s and sanya’s studies most closely related to? daniel’s field of study is related to science, and sanya’s field of study is related to .
Answers: 3

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2
You know the right answer?
What is the molarity of 416.48 g bacl2 dissolved in 2 l of water? ! a) 1 m b) 104.12 m c) 2 m d)...
Questions



Mathematics, 24.10.2019 15:43




English, 24.10.2019 15:43



Mathematics, 24.10.2019 15:43

English, 24.10.2019 15:43


Social Studies, 24.10.2019 15:43

Business, 24.10.2019 15:43

History, 24.10.2019 15:43



Physics, 24.10.2019 15:43


History, 24.10.2019 15:43