Chemistry, 30.06.2019 19:00 lilchannelll4125
Al2(so4)3(s) + h2o(l) al2o3(s) + h2so4 (aq) calculate enthalpy formation for this reaction. balance the reaction. calculate the total enthalpy change that would occur from 15 moles of al2(so4)3

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:50
Nitrogen dioxide reacts with water to form nitric acid and nitrogen monoxide according to the equation: 3no2(g)+h2o(l)→2hno3(l)+no(g) part a suppose that 4.2 mol no2 and 0.50 mol h2o combine and react completely. which reactant is in excess? express your answer as a chemical formula. nothing
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2

You know the right answer?
Al2(so4)3(s) + h2o(l) al2o3(s) + h2so4 (aq) calculate enthalpy formation for this reaction. balance...
Questions

English, 18.11.2020 01:00



History, 18.11.2020 01:00

Chemistry, 18.11.2020 01:00

English, 18.11.2020 01:00

English, 18.11.2020 01:00

History, 18.11.2020 01:00





English, 18.11.2020 01:00

English, 18.11.2020 01:00

Biology, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00




Mathematics, 18.11.2020 01:00



![H_{reaction}^{0}=[H_{f}^{0}(Al_{2}O_{3}(s)) + (3*H_{f}^{0}(H_{2}SO_{4}(aq))] - [H_{f}^{0}(Al_{2}SO_{4}(aq)) + (3*H_{f}^{0}(H_{2}O(l))]](/tpl/images/0035/8926/80689.png)
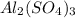 reacts will be=
reacts will be=