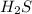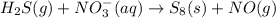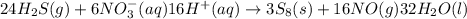Balance the following skeleton reaction and identify the oxidizing and reducing agents: h2s(g) + no3 > ’ s8(s) + no(g) [acidic] include the states for each reactant and product in the balanced chemical equation. do not include the states when identifying the oxidizing and reducing agents.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1
You know the right answer?
Balance the following skeleton reaction and identify the oxidizing and reducing agents: h2s(g) + no...
Questions



Computers and Technology, 21.10.2020 16:01



Mathematics, 21.10.2020 16:01



Computers and Technology, 21.10.2020 16:01

Social Studies, 21.10.2020 16:01

Biology, 21.10.2020 16:01








Mathematics, 21.10.2020 16:01

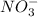 and Reducing agent is
and Reducing agent is