
Chemistry, 30.06.2019 11:00 BeautyxQueen
In the balanced equation , cs2 + 3o2 = co2 + 2so2 , how many mol of o2 would react with 34.5 mol of co2?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1

Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
You know the right answer?
In the balanced equation , cs2 + 3o2 = co2 + 2so2 , how many mol of o2 would react with 34.5 mol of...
Questions







Mathematics, 22.04.2021 04:00




Mathematics, 22.04.2021 04:00



Social Studies, 22.04.2021 04:00

Mathematics, 22.04.2021 04:00

Mathematics, 22.04.2021 04:00

Mathematics, 22.04.2021 04:00


Geography, 22.04.2021 04:00

Mathematics, 22.04.2021 04:00

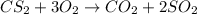
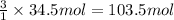 oxygen gas.
oxygen gas.

