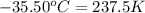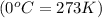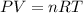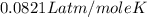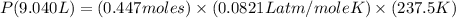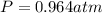Chemistry, 29.06.2019 20:30 cdjeter12oxoait
Agas system has volume, moles and temperature of 9040 ml, 0.447 moles and -35.50 oc, respectively. what is the pressure in atm?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
Agas system has volume, moles and temperature of 9040 ml, 0.447 moles and -35.50 oc, respectively. w...
Questions


Business, 04.08.2021 19:20

English, 04.08.2021 19:20



Chemistry, 04.08.2021 19:20




Chemistry, 04.08.2021 19:30

Computers and Technology, 04.08.2021 19:30

Mathematics, 04.08.2021 19:30



English, 04.08.2021 19:30

English, 04.08.2021 19:30

Mathematics, 04.08.2021 19:30

SAT, 04.08.2021 19:30

English, 04.08.2021 19:30