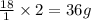Chemistry, 28.06.2019 23:00 mercydiaz84
The following chemical equation describes the chemical reaction of hydrogen gas and oxygen gas to create water 2h2 + o2 = 2h2o use what you know about molar relationships to explain how scientists can predict the amount of water produced if they know the amounts of hydrogen and oxygen gases they have to react.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1
You know the right answer?
The following chemical equation describes the chemical reaction of hydrogen gas and oxygen gas to cr...
Questions


Mathematics, 06.10.2020 14:01

English, 06.10.2020 14:01

Health, 06.10.2020 14:01



Health, 06.10.2020 14:01


Physics, 06.10.2020 14:01

History, 06.10.2020 14:01



Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

Social Studies, 06.10.2020 14:01




Mathematics, 06.10.2020 14:01

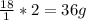 .
.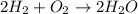
 weighs 2 g.
weighs 2 g.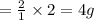
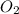 weighs 32 g
weighs 32 g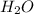 weighs = 18g
weighs = 18g