
Chemistry, 28.06.2019 23:00 teagan1716
How many moles of carbon are in a sample of 21.45 moles of heptane(

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 04:00
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

You know the right answer?
How many moles of carbon are in a sample of 21.45 moles of heptane(...
Questions

History, 27.05.2020 19:00


Social Studies, 27.05.2020 19:00

Mathematics, 27.05.2020 19:00


Mathematics, 27.05.2020 19:00


Mathematics, 27.05.2020 19:00


Mathematics, 27.05.2020 19:00

Physics, 27.05.2020 19:00

Mathematics, 27.05.2020 19:00



English, 27.05.2020 19:00


Chemistry, 27.05.2020 19:00

Mathematics, 27.05.2020 19:00


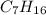
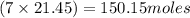 of carbon atom
of carbon atom

