
Chemistry, 28.06.2019 20:00 AlexGaming1822
If you make a solution by dissolving 1.0 mol of fecl3 into 1.0 kg of water, how would the osmotic pressure of this solution compare with the osmotic pressure of a solution that is made from 1.0 mol of glucose in 1.0 kg of water? one-half as large the same twice as large four times as large

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3
You know the right answer?
If you make a solution by dissolving 1.0 mol of fecl3 into 1.0 kg of water, how would the osmotic pr...
Questions


Health, 13.07.2019 01:00


Health, 13.07.2019 01:00





Social Studies, 13.07.2019 01:00

Mathematics, 13.07.2019 01:00



Mathematics, 13.07.2019 01:00



Chemistry, 13.07.2019 01:00

Mathematics, 13.07.2019 01:00

Chemistry, 13.07.2019 01:00


English, 13.07.2019 01:00

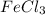 in 1.0 kg water and 1.0 mol glucose in 1.0 kg water.
in 1.0 kg water and 1.0 mol glucose in 1.0 kg water. 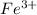 and 3
and 3 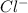 ions.
ions.

