
Ethanol (c2h5oh) melts at -114â°c. the enthalpy of fusion is 5.02 kj/mol. the specific heats of solid and liquid ethanol are 0.97 j/gk and 2.3 j/gk, respectively. how much heat (kj) is needed to convert 25.0 g of solid ethanol at -135â°c to liquid ethanol at -50â°c?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Select all of the statements which are true. electrons are located in shells or orbits around the atom. electrons orbit slowly around the atom. electrons travel in one flat path around the nucleus of an atom. the valence of an atom is determined by the number of electrons in the atom's outermost shell.
Answers: 1



Chemistry, 23.06.2019 12:00
372 ml is the volume of aluminum, density is 2.70 g/ml what is the mass in grams
Answers: 1
You know the right answer?
Ethanol (c2h5oh) melts at -114â°c. the enthalpy of fusion is 5.02 kj/mol. the specific heats of soli...
Questions

Mathematics, 29.10.2020 19:40

Mathematics, 29.10.2020 19:40

Mathematics, 29.10.2020 19:40


Mathematics, 29.10.2020 19:40






Mathematics, 29.10.2020 19:40

Chemistry, 29.10.2020 19:40

Mathematics, 29.10.2020 19:40

Biology, 29.10.2020 19:40

Mathematics, 29.10.2020 19:40


Biology, 29.10.2020 19:40



Geography, 29.10.2020 19:40

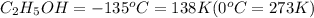
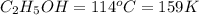
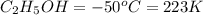
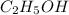 at 159 K
at 159 K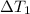 = 159 K - 138 K = 21 K
= 159 K - 138 K = 21 K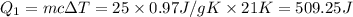
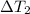 = 223 K - 159 K = 64 K
= 223 K - 159 K = 64 K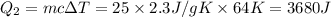
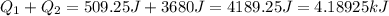 (1kJ=1000J)
(1kJ=1000J)

