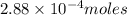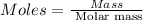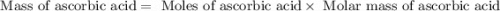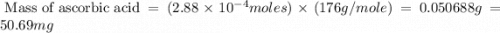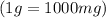Chemistry, 28.06.2019 17:30 kprincess16r
Ascorbic acid, or vitamin c (c6h8o6, molar mass = 176 g/mol), is a naturally occurring organic compound with antioxidant properties. a healthy adult’s daily requirement of vitamin c is 70-90 mg. a sweet lime contains 2.88×10−4 mol of ascorbic acid. to determine whether the ascorbic acid in a sweet lime meets the daily requirement, calculate the mass of ascorbic acid in 2.88×10−4 mol of ascorbic acid.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
What are the major products produced in the combustion of c10h22 under the following conditions? write balanced chemical equations for each. a. an excess of oxygen b. a slightly limited oxygen supply c. a very limited supply of oxygen d. the compound is burned in air
Answers: 2


Chemistry, 21.06.2019 21:30
The density of an unknown gas at 98°c and 740 mmhg is 2.50 g/l. what is the molar mass of the gas with work showed?
Answers: 1

Chemistry, 21.06.2019 22:30
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1
You know the right answer?
Ascorbic acid, or vitamin c (c6h8o6, molar mass = 176 g/mol), is a naturally occurring organic compo...
Questions

Mathematics, 12.07.2019 14:00

Mathematics, 12.07.2019 14:00

Social Studies, 12.07.2019 14:00


Biology, 12.07.2019 14:00

Biology, 12.07.2019 14:00



History, 12.07.2019 14:00

Social Studies, 12.07.2019 14:00

Mathematics, 12.07.2019 14:00

Social Studies, 12.07.2019 14:00


Social Studies, 12.07.2019 14:00


Biology, 12.07.2019 14:00


French, 12.07.2019 14:00


Business, 12.07.2019 14:00