
Chemistry, 28.06.2019 09:00 Zachary4759
The enthalpy change for converting 1.00 mol of ice at -25.0 ∘c to water at 90.0∘c is kj. the specific heats of ice, water, and steam are 2.09 j/g−k, 4.18 j/g−k, and 1.84 j/g−k, respectively. for h2o, δ hfus = 6.01kj/mol, and δhvap = 40.67 kj/mol.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 10:00
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 14:30
1) describe the physical layout of the ocean floor ? 2) explain how the dumbo octopus swims differently than other octopus species and why this would be an advantage in the aphonic zone . 3) why are the types of organisms that live at each underwater hot vent so dramatically different ?
Answers: 3
You know the right answer?
The enthalpy change for converting 1.00 mol of ice at -25.0 ∘c to water at 90.0∘c is kj. the specif...
Questions

Mathematics, 26.09.2019 05:00

Arts, 26.09.2019 05:00

Mathematics, 26.09.2019 05:00

Social Studies, 26.09.2019 05:00

English, 26.09.2019 05:00

English, 26.09.2019 05:00

Mathematics, 26.09.2019 05:00




Biology, 26.09.2019 05:00

Biology, 26.09.2019 05:00

Social Studies, 26.09.2019 05:00


Advanced Placement (AP), 26.09.2019 05:00





Chemistry, 26.09.2019 05:00

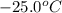 to water at
to water at 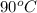 is, 7.712 KJ
is, 7.712 KJ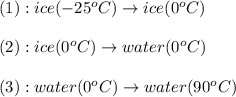
![\Delta H=[m\times c_{ice}\times (T_2-T_1)]+\Delta H_{fusion}+[m\times c_{water}\times (T_3-T_2)]](/tpl/images/0026/6160/eeaad.png)
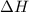 = enthalpy change
= enthalpy change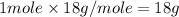
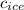 = specific heat of ice = 2.09 J/gk
= specific heat of ice = 2.09 J/gk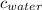 = specific heat of water = 4.18 J/gk
= specific heat of water = 4.18 J/gk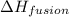 = enthalpy change for fusion = 6.01 KJ/mole = 0.00601 J/mole
= enthalpy change for fusion = 6.01 KJ/mole = 0.00601 J/mole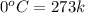
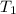 = initial temperature of ice =
= initial temperature of ice = 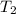 = final temperature of ice =
= final temperature of ice = 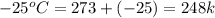
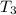 = initial temperature of water =
= initial temperature of water = 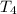 = final temperature of water =
= final temperature of water = 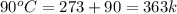
![\Delta H=[18g\times 2.09J/gK\times (273-248)k]+0.00601J+[18g\times 4.18J/gK\times (363-273)k]](/tpl/images/0026/6160/21744.png)
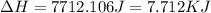 (1 KJ = 1000 J)
(1 KJ = 1000 J)

