

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1


Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1

Chemistry, 23.06.2019 04:00
Why must humans find substitutes for many minerals found on earth? (a) form at an extremely slow rate (b) controlled by other countries (c) too deep in the earth to collect
Answers: 1
You know the right answer?
Balance the equation with the correct coefficients __kcio3 -> __kci +...
Questions


Mathematics, 18.12.2020 08:20


History, 18.12.2020 08:20

Mathematics, 18.12.2020 08:20

Health, 18.12.2020 08:20



Mathematics, 18.12.2020 08:20



Mathematics, 18.12.2020 08:20

Health, 18.12.2020 08:20

Social Studies, 18.12.2020 08:20

Chemistry, 18.12.2020 08:20


Biology, 18.12.2020 08:20



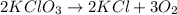
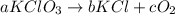
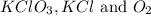 respectively.
respectively.

