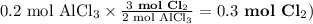Chemistry, 27.06.2019 04:00 tracyaleblanc
*explain your answer for brainliestaluminum reacts with chlorine gas to produce aluminum chloride according to the following equation. al + cl2 → alcl3which of the following fractions can be used for the mole ratio to determine the mass of cl2 from a known mass of alcl3? two over one three over two one over two two over three

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is a scientific theory? a. a scientist's guess about how something works b. the results of an experiment obtained using the scientific method c. a proven fact that will never change d. an idea that is backed by data from many sources
Answers: 2

Chemistry, 22.06.2019 04:40
Listen base your answer to the question on the information below.propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below.c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol.what is the correct structural formula for a molecule of methanethiol
Answers: 3


Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium chlorate
Answers: 3
You know the right answer?
*explain your answer for brainliestaluminum reacts with chlorine gas to produce aluminum chloride ac...
Questions

Mathematics, 16.01.2020 19:31



Physics, 16.01.2020 19:31

Health, 16.01.2020 19:31


Mathematics, 16.01.2020 19:31

Computers and Technology, 16.01.2020 19:31






Biology, 16.01.2020 19:31

Health, 16.01.2020 19:31

Biology, 16.01.2020 19:31


English, 16.01.2020 19:31

Biology, 16.01.2020 19:31

Arts, 16.01.2020 19:31