
Chemistry, 26.06.2019 12:00 mcmccann4317
How many moles of gold are equivalent to 1.204 × 1024 atoms?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:10
Starch and are common polysaccharide carbohydrates found in plants. sucrose glycogen fructose cellulose
Answers: 3

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
How many moles of gold are equivalent to 1.204 × 1024 atoms?...
Questions

History, 02.08.2019 16:30


Biology, 02.08.2019 16:30

Mathematics, 02.08.2019 16:30


History, 02.08.2019 16:30


History, 02.08.2019 16:30

History, 02.08.2019 16:30




Arts, 02.08.2019 16:30

Chemistry, 02.08.2019 16:30



Biology, 02.08.2019 16:30

Mathematics, 02.08.2019 16:30


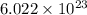 .
.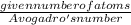
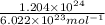
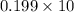 mol
mol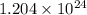 atoms.
atoms.

