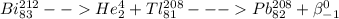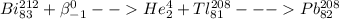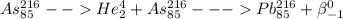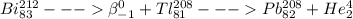Which of the following represents a balanced nuclear equation showing bismuth-212 undergoing alpha decay followed by beta decay? (2 points) superscript 212 over subscript 83 bi yields superscript 4 over subscript 2 he + superscript 208 over subscript 81 tl yields x−superscript 208 over subscript 82 pb + superscript 0 over subscript -1 beta superscript 212 over subscript 83 bi + superscript 0 over subscript -1 beta yields superscript 4 over subscript 2 he + superscript 208 over subscript 81 tl yields x−superscript 208 over subscript 82 pb superscript 216 over subscript 85 as yields superscript 4 over subscript 2 he + superscript 216 over subscript 85 as yields superscript 216 over subscript 85 pb + superscript 0 over subscript -1 beta superscript 212 over subscript 83 bi yields superscript 0 over subscript -1 beta + superscript 208 over subscript 81 tl yields superscript 208 over subscript 82 pb + superscript 4 over subscript 2 he

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1

Chemistry, 22.06.2019 09:30
The chart shows the bid provided by four contractors to complete a job. which contractor is the most cost-effective?
Answers: 3

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1
You know the right answer?
Which of the following represents a balanced nuclear equation showing bismuth-212 undergoing alpha d...
Questions















Computers and Technology, 03.09.2019 18:20



Computers and Technology, 03.09.2019 18:20