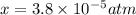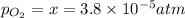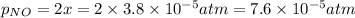Chemistry, 22.06.2019 20:00 denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3
You know the right answer?
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 1...
Questions

Social Studies, 06.05.2020 04:19

Mathematics, 06.05.2020 04:19


Mathematics, 06.05.2020 04:19


History, 06.05.2020 04:19


History, 06.05.2020 04:19




Mathematics, 06.05.2020 04:19


Social Studies, 06.05.2020 04:19


Biology, 06.05.2020 04:19

Chemistry, 06.05.2020 04:19



Computers and Technology, 06.05.2020 04:19

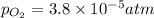
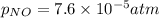
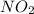 = p = 0.70 atm
= p = 0.70 atm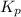 for the reaction
for the reaction 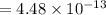
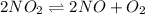
![K_p=\frac{[2x]^2[x]}{[p-2x]^2}](/tpl/images/0005/2998/5610b.png)
![4.48\times 10^{-13}=\frac{4x^3}{[0.70-2x]^2}](/tpl/images/0005/2998/9a087.png)