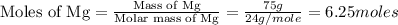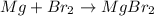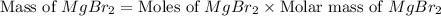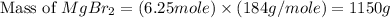Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 20:30
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1

Chemistry, 23.06.2019 00:00
Mercury turns to a vapor at 629.88 k. how much heat is lost when 75.0 g of mercury vapor at 650 k condenses to a liquid at 297 k?
Answers: 1
You know the right answer?
How many grams of mgbr2 are needed to produce 75g or metal?...
Questions

Biology, 12.12.2019 14:31

Mathematics, 12.12.2019 14:31

History, 12.12.2019 14:31

Social Studies, 12.12.2019 14:31

Mathematics, 12.12.2019 14:31

History, 12.12.2019 14:31

Social Studies, 12.12.2019 14:31





Physics, 12.12.2019 14:31


Biology, 12.12.2019 14:31


Mathematics, 12.12.2019 14:31

English, 12.12.2019 14:31

Business, 12.12.2019 14:31


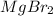 needed to produce are, 1150 grams
needed to produce are, 1150 grams