

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
What is a possible quantum number set for an electron in the 3s orbital of a magnesium atom...
Questions


Computers and Technology, 23.05.2020 19:00


Mathematics, 23.05.2020 19:00





Social Studies, 23.05.2020 19:00


Law, 23.05.2020 19:00




Mathematics, 23.05.2020 19:01

Social Studies, 23.05.2020 19:01

Mathematics, 23.05.2020 19:01



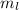 = 0
= 0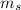 = 1/2 or -1/2Explanation
= 1/2 or -1/2Explanation

