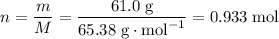Chemistry, 03.02.2020 07:48 diegovaldes25
How many moles of zncl2 will be produced from 61.0g of zn, assuming hcl is available in excess?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2

Chemistry, 23.06.2019 05:00
Which characteristics affect ocean water’s temperature? check all that apply. depth location mass salinity waves
Answers: 1
You know the right answer?
How many moles of zncl2 will be produced from 61.0g of zn, assuming hcl is available in excess?...
Questions

Physics, 05.10.2019 22:20

English, 05.10.2019 22:20


English, 05.10.2019 22:20







Biology, 05.10.2019 22:30

Mathematics, 05.10.2019 22:30





Computers and Technology, 05.10.2019 22:30



Chemistry, 05.10.2019 22:30