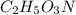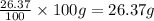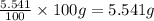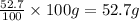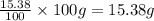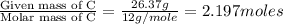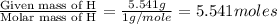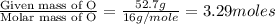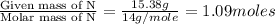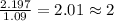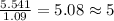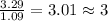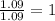Chemistry, 16.11.2019 16:31 gabegabemm1
What is the molecular formula for a compound that is 26.37 carbon 5.541 hydrogen 52.70 oxygen and 15.38 nitrogen and has a molar mass of 182.16g

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1


Chemistry, 22.06.2019 23:30
The sum of the oxidation numbers in a neutral compound is always
Answers: 2
You know the right answer?
What is the molecular formula for a compound that is 26.37 carbon 5.541 hydrogen 52.70 oxygen and 15...
Questions

Mathematics, 26.11.2019 18:31


Business, 26.11.2019 18:31

Mathematics, 26.11.2019 18:31



Mathematics, 26.11.2019 18:31




History, 26.11.2019 18:31






History, 26.11.2019 18:31