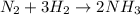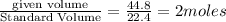Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 09:00
Given the following reaction: c3h8+5o2=3co2+4h20 how many grams of co2 will be produced 7 g of c3h8 and 98 g of o2
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
For the haber process, n2+3h2 yields 2nh3, what volume of nitrogen is consumed at stp if you collect...
Questions

Mathematics, 16.10.2020 08:01


Chemistry, 16.10.2020 08:01

Chemistry, 16.10.2020 08:01

Social Studies, 16.10.2020 08:01




Mathematics, 16.10.2020 08:01



Mathematics, 16.10.2020 08:01

History, 16.10.2020 08:01




Mathematics, 16.10.2020 08:01

History, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01