
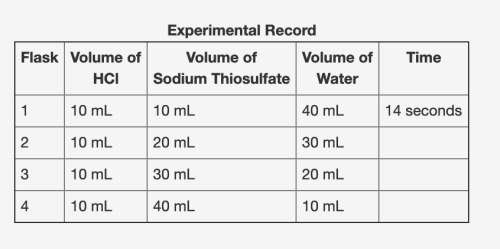
In an experiment, hydrochloric acid reacted with different volumes of sodium thiosulfate in water. a yellow precipitate was formed during the reaction. a cross drawn at the base of each flask became gradually invisible due the formation of this yellow precipitate. the time taken for the cross to become invisible was recorded. a partial record of the experiment is shown.
based on your knowledge of factors that affect the rates of chemical reactions, predict the trend in the last column of the experimental record. use complete sentences to explain the trend you predicted. you do not have to determine exact values for time; just describe the trend you would expect (increase or decrease) and why it occurs.
plz answer asap

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 06:30
Acompound has the molecular formula c3h8. which class of organic compounds does it belong to?
Answers: 1

Chemistry, 23.06.2019 06:30
Acertain atom has 22 protons and 19 electrons. this atom loses an electron. the net charge on the atom is now 4+1+01-4-. if this same atom with 22 protons and 19 electrons were to gain 3 electrons, the net charge on the atom would be 3+2+02-3-.
Answers: 1


Chemistry, 23.06.2019 17:00
Achemist tests new mixtures for paint samples and decides one mixture's stickiness makes it worthless. a scientist working in a nearby lab suggests a different use for this sticky mixture. which statement best describes this situation?
Answers: 1
You know the right answer?
In an experiment, hydrochloric acid reacted with different volumes of sodium thiosulfate in water. a...
Questions

Business, 09.11.2019 01:31


Mathematics, 09.11.2019 01:31

Mathematics, 09.11.2019 01:31




Mathematics, 09.11.2019 01:31



Engineering, 09.11.2019 01:31


Chemistry, 09.11.2019 01:31



Mathematics, 09.11.2019 01:31

Mathematics, 09.11.2019 01:31

Arts, 09.11.2019 01:31




