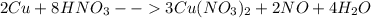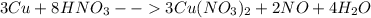Given the reaction below:
2cu + 8hno3 --> 3cu(no3)2 +2no + 4h2o
the mol...

Chemistry, 01.02.2020 17:45 santana647
Given the reaction below:
2cu + 8hno3 --> 3cu(no3)2 +2no + 4h2o
the mole ratio of no produced to hno3 reacted is
a. 4: 1
b. 1: 4
c. 2: 3
d. 2: 3

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1

Chemistry, 22.06.2019 20:30
What is a difference between a mixture of elements and a mixture of compounds
Answers: 1
You know the right answer?
Questions

Mathematics, 27.10.2020 18:00





Mathematics, 27.10.2020 18:00


Computers and Technology, 27.10.2020 18:00

Mathematics, 27.10.2020 18:00



Mathematics, 27.10.2020 18:00

Biology, 27.10.2020 18:00

Social Studies, 27.10.2020 18:00

Chemistry, 27.10.2020 18:00

Mathematics, 27.10.2020 18:00