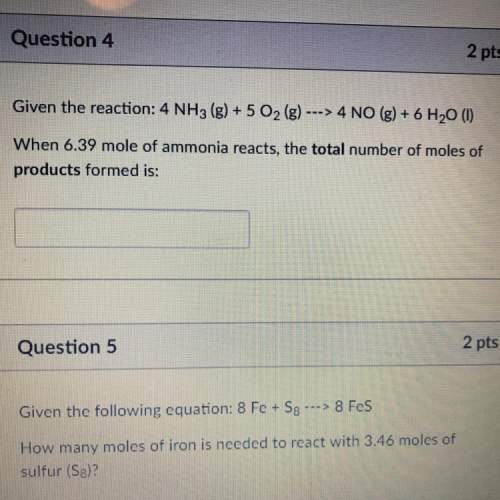
‼️stoichiometry / moles problem: ‼️
...

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
You know the right answer?
Questions



Mathematics, 25.02.2021 03:50


Biology, 25.02.2021 03:50



Mathematics, 25.02.2021 03:50

Social Studies, 25.02.2021 03:50



Biology, 25.02.2021 03:50

Arts, 25.02.2021 03:50

English, 25.02.2021 04:00



Mathematics, 25.02.2021 04:00

Mathematics, 25.02.2021 04:00

Mathematics, 25.02.2021 04:00

Mathematics, 25.02.2021 04:00