Chemistry, 12.12.2019 11:31 calraezaj2020
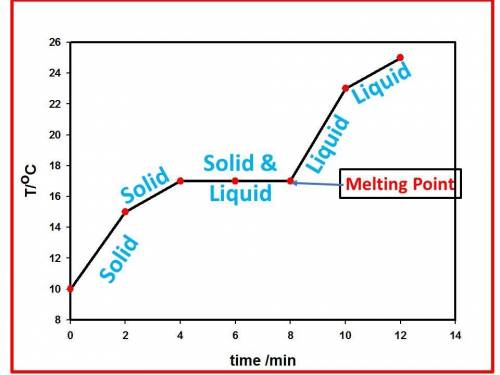
An unknown substance is heated from 10 °c to 25 °c. the table below shows the state of the substance at different temperatures.
time
(minutes) temperature of liquid
(degrees celsius) state of matter
0 10 solid
2 15 solid
4 17
6 17
8 17
10 23 liquid
12 25 liquid
which statement is correct about the unknown substance?
its melting point is 17 °c.
its melting point is 15 °c.
the substance sublimes at 15 °c.
the substance exists as a gas at 17 °c.
(this is science)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Astudent carefully transfers 30 g of water and 30 g of alcohol in a glass tube, forming two layers and filling the tube completely. after sealing the tube, the student mixes the solutions, and notices a bubble that forms in the tube. what is the mass of the contents in the glass tube after mixing?
Answers: 2

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3

Chemistry, 23.06.2019 03:30
The molar mass of iron(fe) is 55.8 g/mol. what is the mass in grams of 2.25 moles of iron?
Answers: 1

Chemistry, 23.06.2019 06:30
Generally, observed behavior that can be formulated into a statement, sometimes mathematical in nature, is called a(n): a. observation. b. measurement. c. theory. d. natural law. e. experiment.
Answers: 2
You know the right answer?
An unknown substance is heated from 10 °c to 25 °c. the table below shows the state of the substance...
Questions

Mathematics, 28.10.2020 18:30



Mathematics, 28.10.2020 18:30






History, 28.10.2020 18:30


Mathematics, 28.10.2020 18:30


Mathematics, 28.10.2020 18:30

Computers and Technology, 28.10.2020 18:30



Computers and Technology, 28.10.2020 18:30

Engineering, 28.10.2020 18:30