
Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
What is the net ion charge of beryllium?...
Questions


History, 09.07.2019 18:30

History, 09.07.2019 18:30


Mathematics, 09.07.2019 18:30

Mathematics, 09.07.2019 18:30

Mathematics, 09.07.2019 18:30

Mathematics, 09.07.2019 18:30



Mathematics, 09.07.2019 18:30

Biology, 09.07.2019 18:30

Mathematics, 09.07.2019 18:30


Mathematics, 09.07.2019 18:30

Computers and Technology, 09.07.2019 18:30

Mathematics, 09.07.2019 18:30

Biology, 09.07.2019 18:30

Mathematics, 09.07.2019 18:30

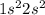 .
.

