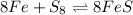Look at the following reaction. select the option that best applies to the reaction.
8fe + s8 ⇄ 8fes
eight moles of reactants form eight moles of product.
nine moles of reactants form eight moles of product.
eight moles of products form eight moles of reactants.
this reaction is not at equilibrium.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2
You know the right answer?
Look at the following reaction. select the option that best applies to the reaction.
8fe + s8...
8fe + s8...
Questions

Chemistry, 12.10.2019 11:00



Mathematics, 12.10.2019 11:00

Mathematics, 12.10.2019 11:00

Social Studies, 12.10.2019 11:00








Arts, 12.10.2019 11:00


Biology, 12.10.2019 11:00