
Chemistry, 15.12.2019 17:31 Bianca1203
Avolume of 25.0 ml of nitric acid, hno3, is titrated with .12 m naoh. to completely neutralize the acid 10.0 ml of naoh must be added. what is the molarity of nitric acid is

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
Avolume of 25.0 ml of nitric acid, hno3, is titrated with .12 m naoh. to completely neutralize the a...
Questions

Mathematics, 16.07.2019 13:30


Mathematics, 16.07.2019 13:30


History, 16.07.2019 13:30


Business, 16.07.2019 13:30

Computers and Technology, 16.07.2019 13:30





History, 16.07.2019 13:30





History, 16.07.2019 13:30

Mathematics, 16.07.2019 13:30

Mathematics, 16.07.2019 13:30

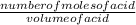
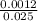 = 0.048M
= 0.048M

