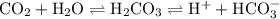Chemistry, 13.01.2020 13:31 Bengynease2598
16. explain hov
to explain how (in terms of a shift in reaction rates in response to putting a chemical stress on the system) the
following equilibrium buffer system in human blood maintain homeostatic ph levels:
co2 + h20 --> h2co3 -> h+ + hco3

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1
You know the right answer?
16. explain hov
to explain how (in terms of a shift in reaction rates in response to putting a...
to explain how (in terms of a shift in reaction rates in response to putting a...
Questions




History, 16.07.2019 07:30

Computers and Technology, 16.07.2019 07:30

Mathematics, 16.07.2019 07:30

Mathematics, 16.07.2019 07:30

Mathematics, 16.07.2019 07:30


History, 16.07.2019 07:30

Mathematics, 16.07.2019 07:30

Geography, 16.07.2019 07:30

Mathematics, 16.07.2019 07:30

Biology, 16.07.2019 07:30



History, 16.07.2019 07:30

Mathematics, 16.07.2019 07:30


Mathematics, 16.07.2019 07:30