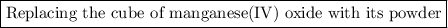Chemistry, 28.06.2019 02:40 MichealM7668
When 1 g of a cube of manganese (iv) oxide was added to 100 cm3 of hydrogen peroxide, water and oxygen were produced. manganese oxide was not used up in the reaction. based on this information, which of the following is likely to increase the rate formation of the products? using 2 grams of manganese oxide using 50 cm3 of hydrogen peroxide replacing the cube of manganese (iv) oxide with its powder removing manganese (iv) oxide from the reaction mixture

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 23:00
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3

Chemistry, 23.06.2019 06:30
Moving force of air flows from areas of high pressure to areas of low pressure true or false
Answers: 2
You know the right answer?
When 1 g of a cube of manganese (iv) oxide was added to 100 cm3 of hydrogen peroxide, water and oxyg...
Questions

Physics, 13.05.2021 14:00

English, 13.05.2021 14:00



Chemistry, 13.05.2021 14:00



History, 13.05.2021 14:00

English, 13.05.2021 14:00




Geography, 13.05.2021 14:00


Chemistry, 13.05.2021 14:00

French, 13.05.2021 14:00


Mathematics, 13.05.2021 14:00


Social Studies, 13.05.2021 14:00