Chemistry, 31.10.2019 07:31 zackarygonzalez1028
Write a balanced equation for the dissolution of caco3? lable all phases so far i have caco3(aq)< --> ca^2+(aq)+co3^2-(aq) but that is wrong.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 12:00
An atom of which element reacts with an atom of hydrogen to form a bond with the greatest degree of polarity ?
Answers: 1

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
Write a balanced equation for the dissolution of caco3? lable all phases so far i have caco3(aq)&l...
Questions


Geography, 09.02.2021 23:00

Biology, 09.02.2021 23:00



Mathematics, 09.02.2021 23:00

Mathematics, 09.02.2021 23:00

Mathematics, 09.02.2021 23:00

English, 09.02.2021 23:00

Chemistry, 09.02.2021 23:00

English, 09.02.2021 23:00


Biology, 09.02.2021 23:00

Mathematics, 09.02.2021 23:00

Mathematics, 09.02.2021 23:00




English, 09.02.2021 23:00

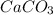 and HCl:
and HCl: