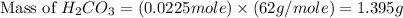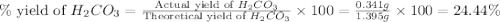Chemistry, 25.06.2019 17:10 sweetbri7p5v6tn
During a laboratory experiment, a 3.81-gram sample of nahco3 was thermally decomposed. in this experiment, carbon dioxide and water vapors escape and are combined to form carbonic acid. after decomposition, the sample weighed 2.86 grams. calculate the percentage yield of carbonic acid for the reaction. describe the calculation process in detail. nahco3 → na2co3 + h2co3

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2



Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2
You know the right answer?
During a laboratory experiment, a 3.81-gram sample of nahco3 was thermally decomposed. in this exper...
Questions

Mathematics, 01.09.2019 01:30

History, 01.09.2019 01:30

Mathematics, 01.09.2019 01:30


Mathematics, 01.09.2019 01:30



Computers and Technology, 01.09.2019 01:30


Mathematics, 01.09.2019 01:30

Mathematics, 01.09.2019 01:30

English, 01.09.2019 01:30



Mathematics, 01.09.2019 01:30

History, 01.09.2019 01:30




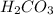 is, 24.44 %
is, 24.44 %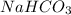 .
.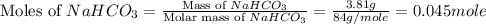
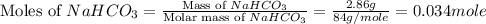
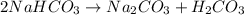
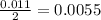 mole of
mole of 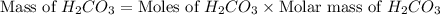
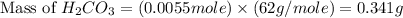
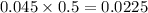 mole of
mole of