Chemistry, 24.06.2019 19:30 edybuza764
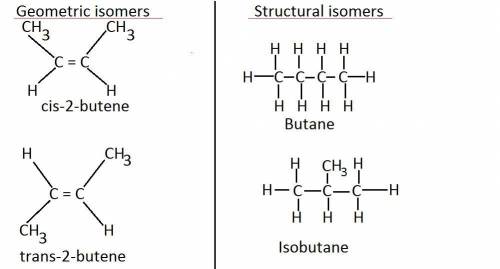
Which isomers can have different physical or chemical properties? structural isomers only geometric isomers only both structural isomers and geometric isomers neither structural isomers nor geometric isomers

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1


Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
Which isomers can have different physical or chemical properties? structural isomers only geometri...
Questions

History, 18.10.2019 19:30

Arts, 18.10.2019 19:30


Mathematics, 18.10.2019 19:30

Mathematics, 18.10.2019 19:30



Mathematics, 18.10.2019 19:30



Mathematics, 18.10.2019 19:30

Arts, 18.10.2019 19:30

Mathematics, 18.10.2019 19:30

Mathematics, 18.10.2019 19:30

Chemistry, 18.10.2019 19:30

Chemistry, 18.10.2019 19:30

History, 18.10.2019 19:30



English, 18.10.2019 19:30