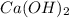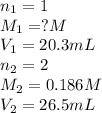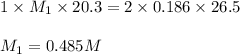Chemistry, 21.06.2019 17:00 sillslola816oxb5h7
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calcium hydroxide. if 26.5 ml of base are required to neutralize 20.3 ml of the acid, what is the molarity of the hydroiodic acid solution? m hydroiodic acid

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 23.06.2019 00:30
Many bird species build nests in which they raise their offspring. one of these species is known as the magnolia warbler. breeding pairs of magnolia warblers build nests out of pieces of grass and twigs. the female warbler will then lay her eggs in the nest and sit on the eggs for a few weeks until they hatch. after the offspring have hatched, the female will continue to sit on the newborn chicks to keep them warm. both the male and the female bring food for the offspring to eat until the young are mature enough to find food on their own. which of these most likely describes why birds such as the magnolia warbler build nests in which to raise their offspring? a. nest building decreases the amount of energy used by the parent birds to raise their offspring to adulthood. b. nest building increases the probability that the offspring will survive and eventually produce offspring of their own. c. nest building decreases the amount of food that the offspring require the adult birds to provide. d. nest building increases the probability of the offspring being located by airborne predators.
Answers: 1

Chemistry, 23.06.2019 01:30
In what way do investigations build scientific knowledge? the results of investigations lead to questions that cannot be tested. they reflect the opinions and social values of scientists, ensuring valid information. the results of investigations lead to new questions, which lead to new investigations. they are not influenced by the research of earlier scientists, so they are able to address gaps in understanding.i
Answers: 1

Chemistry, 23.06.2019 08:10
An experiment is conducted to see if cats preferred skim milk or 2% milk. a cup of skim milkwas put out for 5 kittens and then measured how much the kittens drank over the course of aday. following a cup of 2% milk was purout for the skittens and then masured how much thekittens drank over the course of a day. the same kittens were used and the milk was served atthe same temperature. it was discovered that the cats liked the 2% milk more than the skimmilk. what is the dependent variable in this experiment?
Answers: 1
You know the right answer?
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calci...
Questions


Mathematics, 12.03.2021 08:50

Physics, 12.03.2021 08:50

Mathematics, 12.03.2021 08:50


Mathematics, 12.03.2021 08:50

History, 12.03.2021 08:50

Computers and Technology, 12.03.2021 08:50


Computers and Technology, 12.03.2021 08:50


History, 12.03.2021 08:50

Mathematics, 12.03.2021 08:50


Mathematics, 12.03.2021 08:50


World Languages, 12.03.2021 08:50

Mathematics, 12.03.2021 08:50

Chemistry, 12.03.2021 09:00

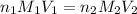
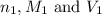 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 
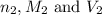 are the n-factor, molarity and volume of base which is
are the n-factor, molarity and volume of base which is