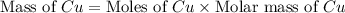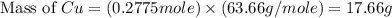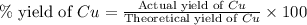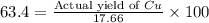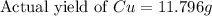Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2


Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1
You know the right answer?
If 5.00 grams of aluminum react with an excess of copper (ii) sulfate and the percentage yield is 63...
Questions







English, 20.09.2020 16:01



Social Studies, 20.09.2020 16:01

History, 20.09.2020 16:01

Mathematics, 20.09.2020 16:01



English, 20.09.2020 16:01

Business, 20.09.2020 16:01


Mathematics, 20.09.2020 16:01


Mathematics, 20.09.2020 16:01

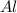 = 5 g
= 5 g
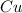 = 63.66 g/mole
= 63.66 g/mole
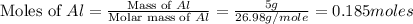
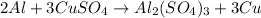
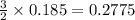 moles of
moles of