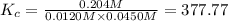Carbonyl chloride (cocl2), also called phosgene, was used in world war i as a poisonous gas. the equilibrium concentrations for the reaction between carbon monoxide and molecular chlorine to form carbonyl chloride at a certain temperature are [co] = 0.0210 m, [cl2] = 0.0450 m, and [cocl2] = 0.204 m. co(g) + cl2(g) ⇆ cocl2(g) calculate the equilibrium constant (kc).

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:00
The overall chemical reaction for photosynthesis isshown below: 6co2 + 6h20 → c6h12o6 + 602what mass of glucose (c6h1206) can form from71.89 g co2? (molar mass of c6h1206 = 180.18g/mol; molar mass of co2 = 44.01 g/mol)71.89 g co2=g c6h1206
Answers: 1

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1


You know the right answer?
Carbonyl chloride (cocl2), also called phosgene, was used in world war i as a poisonous gas. the equ...
Questions


Mathematics, 28.10.2020 03:50





Mathematics, 28.10.2020 03:50

Mathematics, 28.10.2020 03:50


Mathematics, 28.10.2020 03:50

English, 28.10.2020 03:50



Physics, 28.10.2020 03:50



Biology, 28.10.2020 03:50


Mathematics, 28.10.2020 03:50

Mathematics, 28.10.2020 03:50

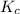
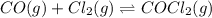
![[CO] = 0.0210 M](/tpl/images/0028/7944/23f4d.png)
![[Cl_2] = 0.0450 M](/tpl/images/0028/7944/558aa.png)
![[COCl_2] = 0.204 M](/tpl/images/0028/7944/1309b.png)
![K_c=\frac{[COCl_2]}{[CO][Cl_2]}](/tpl/images/0028/7944/36d91.png)