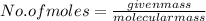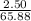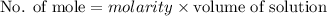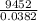Chemistry, 29.06.2019 00:20 webbjalia04
A2.50 g sample of powdered zinc is added to 100.0 ml of a 2.00 m aqueous solution of hydrobromic acid in a calorimeter. the total heat capacity of the calorimeter and solution is 448 j/k. the observed increase in temperature is 21.1 k at a constant pressure of one bar. calculate the standard enthalpy of reaction using these data. zn(s)+2hbr(aq)⟶znbr2(aq)+h2(g)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 1

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2
You know the right answer?
A2.50 g sample of powdered zinc is added to 100.0 ml of a 2.00 m aqueous solution of hydrobromic aci...
Questions

Chemistry, 30.06.2019 13:40

Mathematics, 30.06.2019 13:40

Mathematics, 30.06.2019 13:40

English, 30.06.2019 13:40



Mathematics, 30.06.2019 13:40



Mathematics, 30.06.2019 13:40

Mathematics, 30.06.2019 13:40

Computers and Technology, 30.06.2019 13:40


Physics, 30.06.2019 13:40