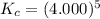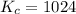Chemistry, 09.07.2019 03:20 chrismcb875
Determine the value of kc for the following reaction if the equilibrium concentrations are as follows: [p4o10]eq = 2.000 moles, [p4]eq = 3.000 moles, [o2]eq = 4.000 m p4o10(s) ↔ p4(s) + 5 o2(g)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Use examples from the article to explain one positive and one negative effect that chemistry has had on society
Answers: 2


Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3
You know the right answer?
Determine the value of kc for the following reaction if the equilibrium concentrations are as follow...
Questions

Biology, 06.07.2019 21:40

Biology, 06.07.2019 21:40

Biology, 06.07.2019 21:40


Social Studies, 06.07.2019 21:40

Social Studies, 06.07.2019 21:40

Social Studies, 06.07.2019 21:40


History, 06.07.2019 21:40


Biology, 06.07.2019 21:40

English, 06.07.2019 21:40


History, 06.07.2019 21:40

Arts, 06.07.2019 21:40

Geography, 06.07.2019 21:40

World Languages, 06.07.2019 21:40



English, 06.07.2019 21:40

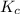 for the following reaction will be, 1024
for the following reaction will be, 1024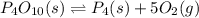
![K_c=[O_2]^5](/tpl/images/0067/9324/32ee9.png)