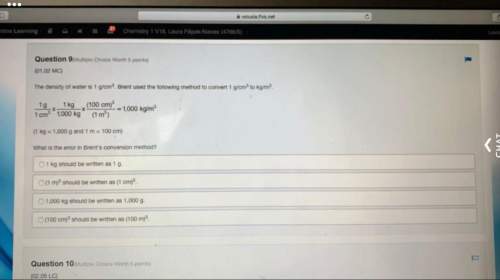
Chemistry, 11.07.2019 22:30 ebonsell4910
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with solution. calculate the volume of solution the student will need to add to reach the final equivalence point. round your answer to significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1


Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 22.06.2019 21:30
While in europe, if you drive 125 km per day, how much money would you spend on gas in one week if gas costs 1.10 euros per liter and your car’s gas mileage is 32.0 mi/gal? assume that 1 euro=1.26 dollars
Answers: 2
You know the right answer?
Achemistry student weighs out of phosphoric acid , a triprotic acid, into a volumetric flask and dil...
Questions

Biology, 03.02.2020 02:49


Mathematics, 03.02.2020 02:49

History, 03.02.2020 02:49


Social Studies, 03.02.2020 02:49


Mathematics, 03.02.2020 02:49


Mathematics, 03.02.2020 02:49

History, 03.02.2020 02:49




Chemistry, 03.02.2020 02:49

Geography, 03.02.2020 02:49

English, 03.02.2020 02:49

History, 03.02.2020 02:49


Mathematics, 03.02.2020 02:49