
Chemistry, 14.07.2019 18:10 Natavia3402
Find the enthalpy of neutralization of hcl and naoh. 137 cm3 of 2.6 mol dm-3 hydrochloric acid was neutralized by 137 cm3 of 2.6 mol dm-3 naoh. the temperature rose from 298 k to 325.8 k. the specific heat capacity is the same as water, 4.18 j/k g.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 15:20
Which description best characterizes the motion of particles in a solid?
Answers: 2

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
You know the right answer?
Find the enthalpy of neutralization of hcl and naoh. 137 cm3 of 2.6 mol dm-3 hydrochloric acid was n...
Questions



Social Studies, 25.02.2021 19:50


English, 25.02.2021 19:50


Mathematics, 25.02.2021 19:50


Arts, 25.02.2021 19:50


Spanish, 25.02.2021 19:50


Business, 25.02.2021 19:50





Mathematics, 25.02.2021 19:50


Biology, 25.02.2021 19:50

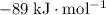 .
.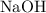 formula units will neutralize one mole of
formula units will neutralize one mole of 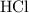 to produce one mole of water.
to produce one mole of water. 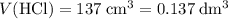 .
.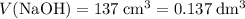 .
.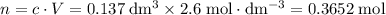 .
.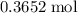 of
of 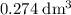 . Assume that the density of the solution is equal to that of water under room temperature.
. Assume that the density of the solution is equal to that of water under room temperature. 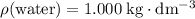 . The mass of the liquid will be
. The mass of the liquid will be 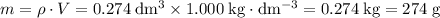 .
.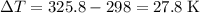 .
.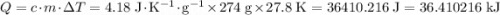 .
. .
.

