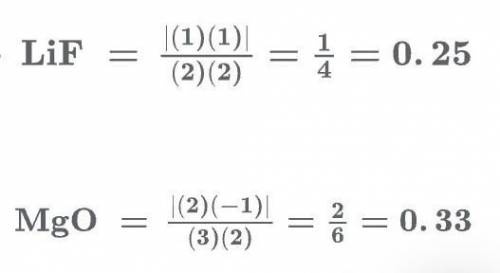Chemistry, 15.07.2019 22:30 marialandingin7520
Which pair of compounds is listed in order of increasing (more exothermic) lattice energy? mgo < cscl lif < lii v lif < mgo cao < bacl2 nacl < kcl flag this question

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 10:40
Asolid that forms and separates from a liquid mixture is called
Answers: 2
You know the right answer?
Which pair of compounds is listed in order of increasing (more exothermic) lattice energy? mgo <...
Questions

Mathematics, 20.05.2020 22:06

History, 20.05.2020 22:06


Health, 20.05.2020 22:06

Biology, 20.05.2020 22:06




History, 20.05.2020 22:06


Biology, 20.05.2020 22:06

Computers and Technology, 20.05.2020 22:06



English, 20.05.2020 22:06

Mathematics, 20.05.2020 22:06

Mathematics, 20.05.2020 22:06

Chemistry, 20.05.2020 22:06

English, 20.05.2020 22:06

Physics, 20.05.2020 22:06