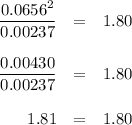Phosphorus pentachloride decomposes according to the chemical equation pcl5(g)↽−−⇀pcl3(g)+cl2(=1.80 at 250 ∘c a 0.197 mol sample of pcl5(g) is injected into an empty 2.90 l reaction vessel held at 250 ∘c. calculate the concentrations of pcl5(g) and pcl3(g) at equilibrium.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3


Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
Phosphorus pentachloride decomposes according to the chemical equation pcl5(g)↽−−⇀pcl3(g)+cl2(=1.80...
Questions

Mathematics, 26.02.2021 17:20

Computers and Technology, 26.02.2021 17:20





Advanced Placement (AP), 26.02.2021 17:20

Mathematics, 26.02.2021 17:20

Mathematics, 26.02.2021 17:20

Mathematics, 26.02.2021 17:20





Arts, 26.02.2021 17:20

Chemistry, 26.02.2021 17:20


Physics, 26.02.2021 17:20


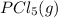 is 0.0655 M and
is 0.0655 M and 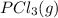 is 0.00240 M at equilibrium.
is 0.00240 M at equilibrium.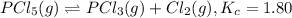
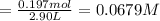

![[PCl_3]=x](/tpl/images/0128/9812/3c44a.png)
![[Cl_2] = x](/tpl/images/0128/9812/a7a23.png)
![=[PCl_5]= (0.0697- x)](/tpl/images/0128/9812/ead0f.png)
![K_c=\frac{[PCl_3][Cl_2]}{[PCl_5]}\\\\1.80=\frac{x\times x}{(0.0679-x)}\\\\x = 0.0655](/tpl/images/0128/9812/79ef0.png)
![=[PCl_5]= (0.0679- x) = (0.0679 -0.0655 )M=0.00240 M](/tpl/images/0128/9812/ac2f8.png)
![= [Cl_2] = x = 0.0655 M](/tpl/images/0128/9812/6faf5.png)
![\text{[PCl$_{5}$]} = \dfrac{\text{0.197 mol}}{\text{2.90 L}} = \text{0.067 93 mol/L}\\\\](/tpl/images/0128/9812/66356.png)
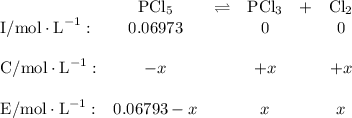
![K_{\text{c}} = \dfrac{\text{[PCl$_3$][Cl$_2$]}}{\text{[PCl$_5$]}} = \dfrac{x^{2}}{0.06793-x} = 1.80\\\\\begin{array}{rcl}\\x^{2}& = & 1.80(0.06793 - x)\\x^{2& = & 0.1223 - 1.80x\\x^{2} + 1.80x - 0.1223& = & 0\\x & = & \mathbf{0.0656}\\\end{array}](/tpl/images/0128/9812/cf1bb.png)