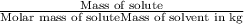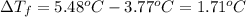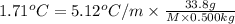Chemistry, 25.07.2019 00:30 allstar976
When a 33.8-g sample of an unknown compound is dissolved in 500. g of benzene, the freezing point of the resulting solution is 3.77°c. the freezing point of pure benzene is 5.48°c, and kf for benzene is 5.12°c/m. calculate the molar mass of the unknown compound.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Calculate the molar mass of aluminum oxide (al2o3). express your answer to four significant figures.
Answers: 1

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 12:30
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3
You know the right answer?
When a 33.8-g sample of an unknown compound is dissolved in 500. g of benzene, the freezing point of...
Questions





Mathematics, 22.08.2019 22:20

Advanced Placement (AP), 22.08.2019 22:20



English, 22.08.2019 22:20








Computers and Technology, 22.08.2019 22:20

Mathematics, 22.08.2019 22:20


Chemistry, 22.08.2019 22:20

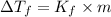
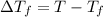
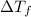 = change in boiling point = 0.81 K
= change in boiling point = 0.81 K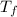 =Boiling point of the solution = 3.77°C
=Boiling point of the solution = 3.77°C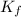 = freezing point constant = 5.12°C/m
= freezing point constant = 5.12°C/m