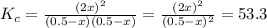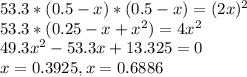Chemistry, 29.07.2019 21:10 sreeranjanig
At a certain temperature, the equilibrium constant, , for this reaction is 53.3. h2(g)+i2(g)↽−−⇀2hi(=53.3 at this temperature, 0.500 mol h2 and 0.500 mol i2 were placed in a 1.00 l container to react. what concentration of hi is present at equilibrium?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 02:30
At 40 âc the solution has at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.g of kno3 per 100 g of water and it can contain up to at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.g of kno3 per 100 g of water. at 0 âc the solubility is ~ at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.kno3 per 100 g of water, so ~ at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.gkno3 per 100 g of water will precipitate out of solution. a kno3 solution containing 55 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 2


Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1
You know the right answer?
At a certain temperature, the equilibrium constant, , for this reaction is 53.3. h2(g)+i2(g)↽−−⇀2hi(...
Questions




History, 12.07.2019 17:30




Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30


Biology, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

Biology, 12.07.2019 17:30

Mathematics, 12.07.2019 17:30

![K_{c}=\frac{[HI]^{2}}{[I_{2}][H_{2}]}](/tpl/images/0148/0680/ff3a2.png)
![[HI]={[HI]}_{0}+2x\\{[H_{2}]}={[H_{2}]}_{0}-x\\{[I_{2}]}={[I_{2}]}_{0}-x\\](/tpl/images/0148/0680/e5b4e.png)