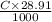Chemistry, 31.07.2019 17:20 drealtania21
Enter your answer in the provided box. sodium hydroxide is used extensively in acid-base titrations because it is a strong, inexpensive base. a sodium hydroxide solution was standardized by titrating 28.58 ml of 0.1851 m standard hydrochloric acid. the initial buret reading of the sodium hydroxide was 1.98 ml, and the final reading was 30.89 ml. what was the molarity of the base solution?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 11:00
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced.be sure your answer has the correct number of significant digits.
Answers: 2

Chemistry, 22.06.2019 19:30
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1

Chemistry, 23.06.2019 05:00
Question 5 match each term to its description. match term definition excess reactant a) reactant that can produce a lesser amount of the product limiting reactant b) reactant that can produce more of the product theoretical yield c) amount of product predicted to be produced by the given reactants
Answers: 2
You know the right answer?
Enter your answer in the provided box. sodium hydroxide is used extensively in acid-base titrations...
Questions



Mathematics, 05.05.2021 23:00

Mathematics, 05.05.2021 23:00






English, 05.05.2021 23:00

Mathematics, 05.05.2021 23:00


Chemistry, 05.05.2021 23:00

Mathematics, 05.05.2021 23:00


French, 05.05.2021 23:00

Mathematics, 05.05.2021 23:00

Social Studies, 05.05.2021 23:00



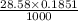 moles
moles