
The decomposition of hydrogen peroxide follows first order kinetics and has a rate constant of 2.54 x 10-4 s-1 at a certain temperature. if the concentration of hydrogen peroxide is 0.321 m after 855 s , what was the initial concentration of hydrogen peroxide at this temperature? 0.258 m0.399 m0.538 m0.677 m1.48 m

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:50
2points why do scientists need governmental funding? o a. government politicians ask all the important scientific questions. o b. scientists have to pay taxes to the government on the money they make. o c. the cost of doing scientific research can be very high. o d. the government is controlled by scientists. submit
Answers: 3

Chemistry, 22.06.2019 03:00
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 23.06.2019 01:00
Which is true concerning the products and reactants of photosynthesis and cellular respiration? a. the products of photosynthesis are sugars and the reactants of cellular respiration are starches. b. the products of photosynthesis are reactants in cellular respiration. c. oxygen is needed for photosynthesis and is given off in cellular respiration.
Answers: 2
You know the right answer?
The decomposition of hydrogen peroxide follows first order kinetics and has a rate constant of 2.54...
Questions




Mathematics, 17.07.2019 01:50

English, 17.07.2019 01:50

Mathematics, 17.07.2019 01:50



History, 17.07.2019 01:50


English, 17.07.2019 01:50

Mathematics, 17.07.2019 01:50

Mathematics, 17.07.2019 01:50





Mathematics, 17.07.2019 01:50

Biology, 17.07.2019 01:50

English, 17.07.2019 01:50

![k=\frac{2.303}{t}\log\frac{[A_o]}{[A]}](/tpl/images/0155/7548/f1041.png)
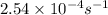
![[A_o]](/tpl/images/0155/7548/dc622.png) = initial amount of the reactant = ?
= initial amount of the reactant = ?![2.54\times 10^{-4}s^{-1}=\frac{2.303}{855s}\log \frac{[A_o]}{0.321}](/tpl/images/0155/7548/e899e.png)
![[A_o]=0.399M](/tpl/images/0155/7548/ee654.png)


