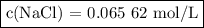Chemistry, 07.08.2019 01:10 BrainlyAvenger
To study the effect of dissolved salt on the rusting of an iron sample, a student prepared a solution of nacl by dissolving 3.038 g of nacl in enough
water to make 792.2 ml of solution. what is the molarity of this solution?
c(nacl)=

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 14:00
Which of the following is true about a carbonated soft drink? . the carbon dioxide is the solvent, and water is the solute.. the water is the solution, and carbon dioxide is the solvent.. the carbon dioxide is the solution, and the water is the solvent.. the water is the solvent, and the carbon dioxide is the solute.. .
Answers: 1

Chemistry, 22.06.2019 17:30
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1
You know the right answer?
To study the effect of dissolved salt on the rusting of an iron sample, a student prepared a solutio...
Questions






Social Studies, 09.10.2019 18:30

History, 09.10.2019 18:30

History, 09.10.2019 18:30

Mathematics, 09.10.2019 18:30





Biology, 09.10.2019 18:30


Mathematics, 09.10.2019 18:30

Social Studies, 09.10.2019 18:30



English, 09.10.2019 18:30