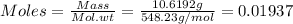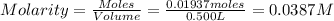Astandard solution is prepared by dissolving 10.6192 g of (nh4)2ce(no3)6 (548.23 g•mol-1, 98.75% purity) in dilute sulfuric acid. the resulting solution is quantitatively transferred to a 500.0-ml volumetric flask and diluted to the mark. what is the ce concentration in the final solution?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
Astandard solution is prepared by dissolving 10.6192 g of (nh4)2ce(no3)6 (548.23 g•mol-1, 98.75% pur...
Questions



English, 14.11.2020 14:00

Geography, 14.11.2020 14:00

Mathematics, 14.11.2020 14:00

History, 14.11.2020 14:00

English, 14.11.2020 14:00

World Languages, 14.11.2020 14:00

English, 14.11.2020 14:00



Spanish, 14.11.2020 14:00


Mathematics, 14.11.2020 14:00


Physics, 14.11.2020 14:00

Physics, 14.11.2020 14:00

History, 14.11.2020 14:00

Mathematics, 14.11.2020 14:00