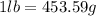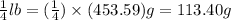Chemistry, 12.08.2019 18:20 marykatherine
To prepare for a laboratory period, a student lab assistant needs 125 g of a compound. a bottle containing 1/4 ib is available. did the student have enough of the compound?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 22:30
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1

Chemistry, 23.06.2019 00:10
Find the missing probability in the table below a.0.10 b.40 c.0.80 d. 0.20
Answers: 2
You know the right answer?
To prepare for a laboratory period, a student lab assistant needs 125 g of a compound. a bottle cont...
Questions


History, 04.08.2019 04:00



Mathematics, 04.08.2019 04:00

Mathematics, 04.08.2019 04:00


Physics, 04.08.2019 04:00

Social Studies, 04.08.2019 04:00



English, 04.08.2019 04:00





Mathematics, 04.08.2019 04:00


History, 04.08.2019 04:00