
Chemistry, 13.08.2019 03:30 armonilucky11
Balance each of the following redox reactions occurring in basic solution. mno−4(aq)+br−(aq)→mno2(s)+bro−3(aq) express your answer as a chemical equation. identify all of the phases in your answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Elements that do not have full outer electron shells will donate, share, or take electrons from other atoms. choose the items that have the correct binary ionic formula.
Answers: 2

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3

Chemistry, 23.06.2019 04:00
How many liters of water can be produced from 5.0liters of butane gas at stp, assuming excess oxygen? c4h10(g) + 02(g) → co2 (e) + h2o (g)
Answers: 2
You know the right answer?
Balance each of the following redox reactions occurring in basic solution. mno−4(aq)+br−(aq)→mno2(s)...
Questions


Chemistry, 16.10.2020 09:01

English, 16.10.2020 09:01







Chemistry, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01


Biology, 16.10.2020 09:01



History, 16.10.2020 09:01


Mathematics, 16.10.2020 09:01


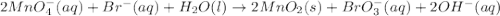
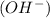 at that side where the less number of hydrogen are present.Now balance the charge.
at that side where the less number of hydrogen are present.Now balance the charge.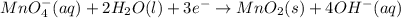 ......(1)
......(1)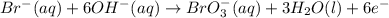 .......(2)
.......(2)

